Table Of Content
- Case‐control studies based within a defined cohort
- Phase III
- High risk, high benefit trial
- Autologous HER2-specific CAR T cells after lymphodepletion for advanced sarcoma: a phase 1 trial
- False-positive errors
- Bispecific CAR T cell therapy targeting BCMA and CD19 in relapsed/refractory multiple myeloma: a phase I/II trial
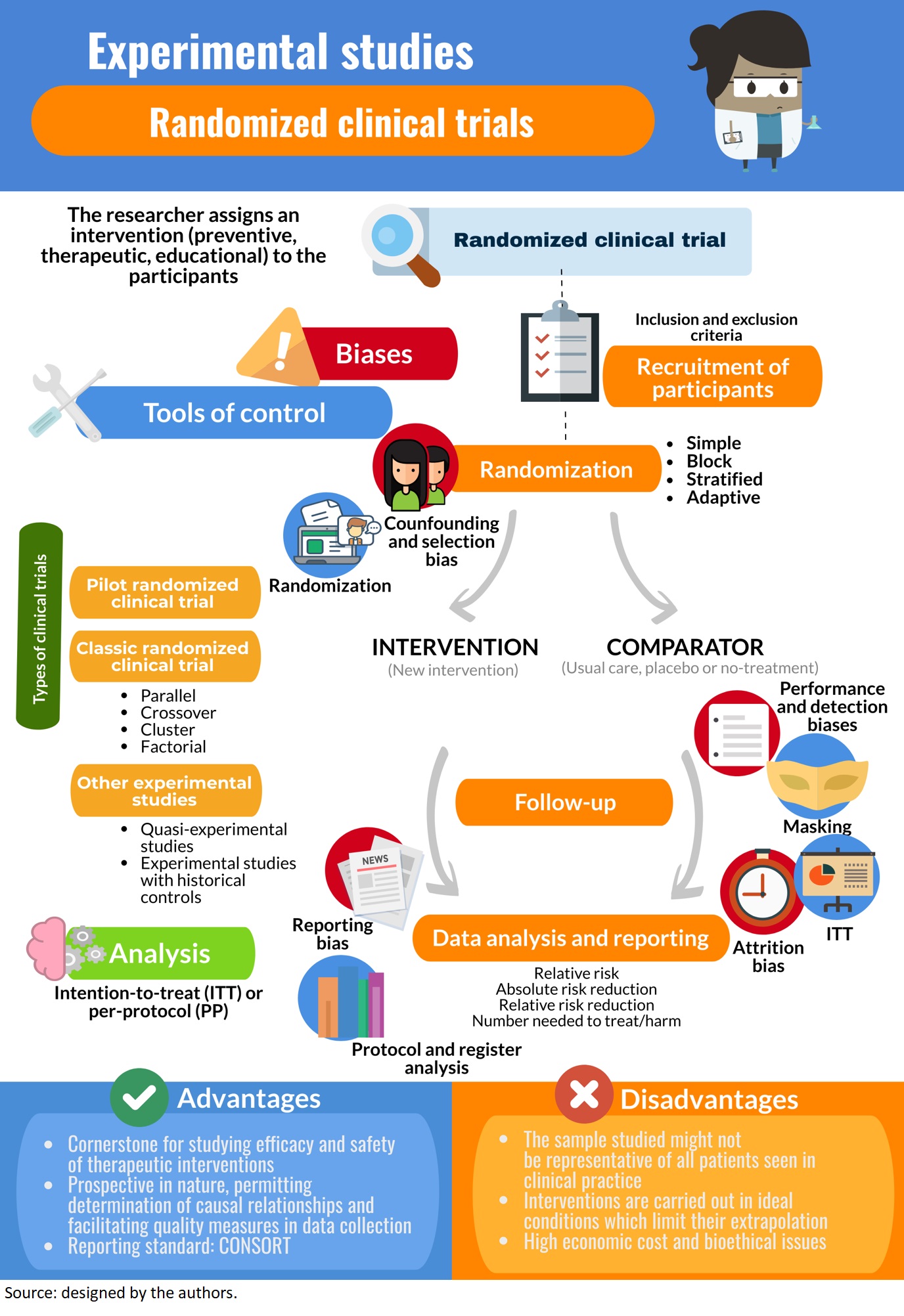
Hence, the outcomes of the intervention/experiment will need to be reversible as this type of study design would not be possible if the subject is undergoing a surgical procedure. A nested case‐control study consists of defining a cohort with suspected risk factors and assigning a control within a cohort to the subject who develops the disease.10 Over a period, cases and controls are identified and followed as per the investigator's protocol. Hence, the case and control are matched on calendar time and length of follow‐up. When this study design is implemented, it is possible for the control that was selected early in the study to develop the disease and become a case in the latter part of the study. Clinical trials, when successful, advance science by providing important insights into disease and treatment mechanisms, eventually changing clinical practice. The results of a trial should be interpretable, whether they are positive or negative; if they are not, then the preliminary data should be reassessed and a new research question considered.
Case‐control studies based within a defined cohort
One disadvantage to the use of placebos is that sometimes they can be costly to obtain. Trials commonly employ stratified randomization to ensure that treatment groups are balanced with respect to confounding variables. In stratified randomization, separate randomization schedules are prepared for each stratum. For example, gender is a potential confounder for estimating the effects of interventions to treat or prevent stroke (e.g., a between-group imbalance with respect to gender could distort the estimate of the intervention effect).
Phase III
Composite endpoints may also reduce the bias due to competing risks and informative censoring. This is because one event can censor other events and if data were only analyzed on a single component then informative censoring can occur. Composite endpoints may also help avoid the multiplicity issue of evaluating many endpoints individually. Historically controlled studies can be considered as a subtype of non‐randomized clinical trial.
High risk, high benefit trial
Based on the above results, in 1795 lemon juice was made a required part of the diet of sailors. Thus, clinical trials can be used to evaluate new therapies, such as new drug or new indication, new drug combination, new surgical procedure or device, new dosing schedule or mode of administration, or a new prevention therapy. One tool to overcome this problem is demonstrated by Griffin and Tsao, who proposed a “mechanism-based classification of phantom limb pain” based on theorized mechanisms of action.
Researchers need to design, conduct, and report on clinical trials to create impactful technologies that can better our understanding of health and medicine. From learning frameworks and reporting methods to determining how to accurately collect, manage, and measure data, you’ll gain the knowledge necessary to understand the components that go into running a successful clinical trial. When constructing entry criteria, the safety of the study participant is paramount. Researcher should consider the appropriateness of recruiting participants with various conditions into the trial. The ability to accrue study participants can also affect the selection of entry criteria.

The advantage with this methodology is that it enables comparability between experiment/intervention groups and thus makes result analysis more efficient. But, with this methodology the covariates will need to be measured and determined before the randomization process. The sample size will help determine the number of strata that would need to be chosen for a study.
The argument framework is a flexible approach to evidence in healthcare
Clinical trials are also known as therapeutic trials, which involve subjects with disease and are placed in different treatment groups. One of the earliest clinical trial studies was performed by James Lind et al in 1747 on sailors with scurvy.12 Lind divided twelve scorbutic sailors into six groups of two. The group who ate two oranges and one lemon had shown the most sudden and visible clinical effects and were taken back at the end of 6 days as being fit for duty. During Lind's time, this was not accepted but was shown to have similar results when repeated 47 years later in an entire fleet of ships.
Dr Dreicer on Improving Clinical Trial Design in Prostate Cancer - OncLive
Dr Dreicer on Improving Clinical Trial Design in Prostate Cancer.
Posted: Fri, 08 Mar 2024 08:00:00 GMT [source]
False-positive errors
The risks inherent in such trials must be minimized, e.g. by cautiously choosing eligibility criteria and stringently monitoring for adverse events. A single outcome should be defined a priori, with sample size and power calculations based on that outcome. Not having a clear predefined primary outcome may lead to an underpowered study, and having multiple outcomes may lead to biased results. Importantly, multiple outcome analyses at a set p value have an increasing risk of type I error (false positives), so the multiplicity should be adjusted for statistically or the analysis will be invalid. If a trial has only one primary endpoint and only one analysis of that endpoint is done the alpha level for that one test will match the overall false positive rate for the trial.
The interventions evaluated can be drugs, devices (e.g., hearing aid), surgeries, behavioral interventions (e.g., smoking cessation program), community health programs (e.g. cancer screening programs) or health delivery systems (e.g., special care units for hospital admissions). We consider clinical trials experiments because the investigators rather than the patients or their doctors select the treatment the patients receive. Results from randomized clinical trials are usually considered the highest level of evidence for determining whether a treatment is effective because trials incorporates features to ensure that evaluation of the benefits and risks of treatments are objective and unbiased. The FDA requires that drugs or biologics (e.g., vaccines) are shown to be effective in clinical trials before they can be sold in the US.
Several features of the I-SPY COVID trial may provide lessons that could be useful beyond the current pandemic. Randomized clinical trials evaluating whether one therapy is better than another nearly always analyze results using the intent-to-treat (ITT) principle. Subjects are analyzed in their assigned/randomized cohort regardless of the intervention they received. ITT analyses are considered “conservative” in that subjects receiving the alternative (non-assigned) intervention skew or “bias” towards showing no difference between the cohorts. In this instance an ITT analysis may result in the incorrect conclusion an intervention is ineffective, a false-negative.
In addition to academic research hospitals, we intentionally recruited community-based sites that do not traditionally participate in clinical trials to enhance enrollment of a broad population. Clinical trials, especially randomized controlled trials, are typically designed to facilitate straightforward interpretation [1]. However, despite randomization, a formal protocol document and clinical trials registries such as clinicaltrials.gov, it remains challenging to appropriately evaluate reports of clinical trials. Herein, we review several issues regarding critical interpretation of clinical trial results. A well-designed trial’s primary endpoint (outcome) should teach you something about the therapeutic process, how well it works (if at all) and its underlying mechanisms.

No comments:
Post a Comment