Table Of Content

Regression models provide estimates of effect sizes (e.g., odds ratios or hazard ratios), which are important when interpreting the results of trials. In addition, regression analyses allow for adjustments for co-variates not used in randomization stratification. Although randomization is likely to balance most factors across arms it does not guarantee balance without stratification. Regression analyses can allow for more precise estimation of effect sizes when there is an imbalance in a prognostic co-variate across arms. Subjects of psoriasis vulgaris are initiated on a biological and a group of patients attain PASI 75 response at 16 weeks.
Avoiding designing failed clinical trials: main issues that lead to invalid data

However, certain treatments cannot be blinded such as surgeries or if the treatment group requires an assessment of the effect of intervention such as quitting smoking. The trial combines phases I/II/III with a dose escalation phase, followed by a pivotal phase at the selected dose. The trial has been approved by French and UK health authorities and includes ambulant boys aged 6 to 10 suffering from Duchenne Muscular Dystrophy.
Clinical trial phases: medical device studies
CDER Launches a Center for Clinical Trial Innovation - FDA.gov
CDER Launches a Center for Clinical Trial Innovation.
Posted: Mon, 15 Apr 2024 07:00:00 GMT [source]
The basic concept of experimental study design is to study the effect of an intervention. In this study design, the risk factor/exposure of interest/treatment is controlled by the investigator. Therefore, these are hypothesis testing studies and can provide the most convincing demonstration of evidence for causality. As a result, the design of the study requires meticulous planning and resources to provide an accurate result. From an epidemiological standpoint, there are two major types of clinical study designs, observational and experimental.3 Observational studies are hypothesis‐generating studies, and they can be further divided into descriptive and analytic. Descriptive observational studies provide a description of the exposure and/or the outcome, and analytic observational studies provide a measurement of the association between the exposure and the outcome.
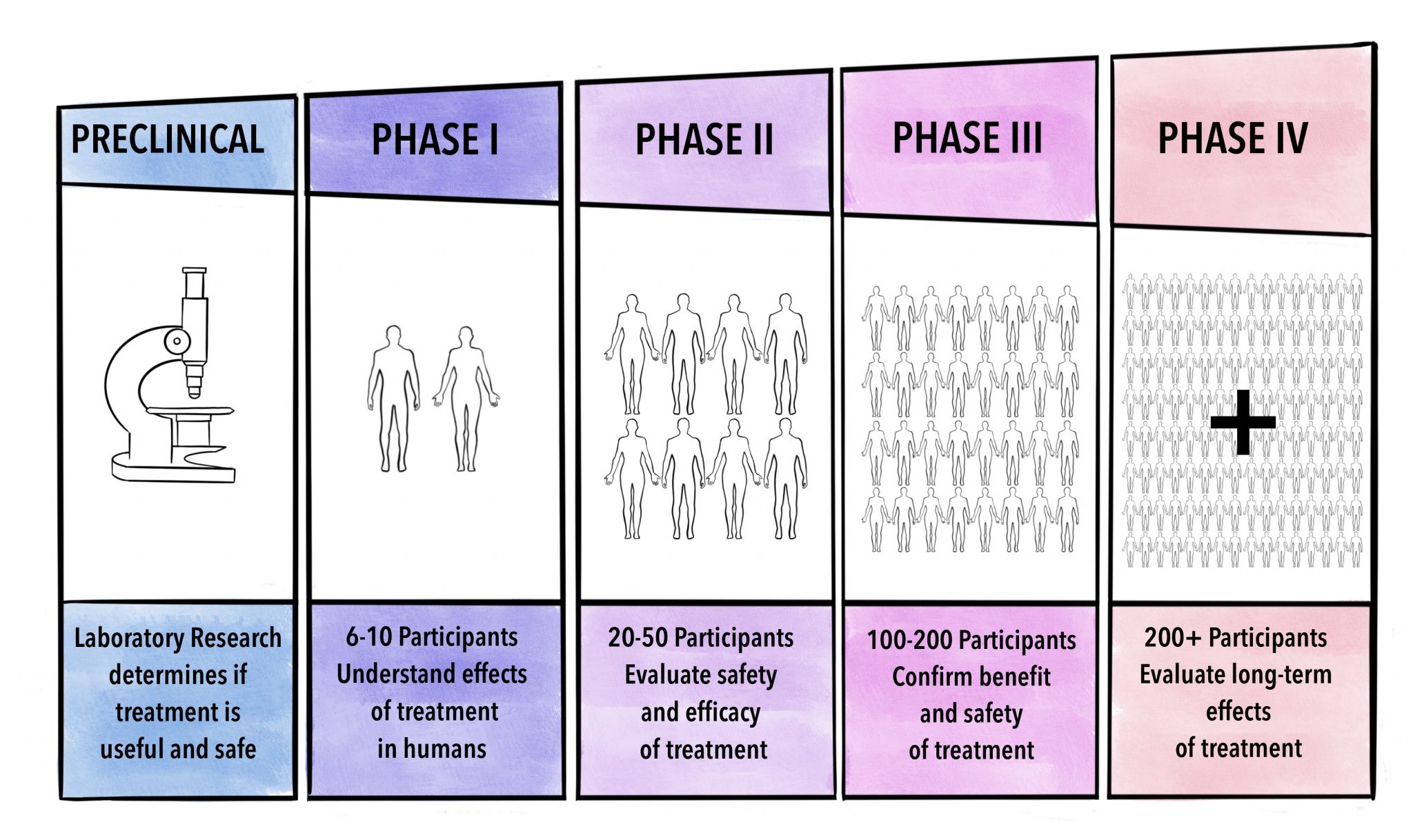
Trial phase:
As chaotic as the early pandemic was, it also spurred tremendous innovation in clinical trial design. The RECOVERY adaptive platform trial in the United Kingdom demonstrated the substantial value of pragmatic phase 3 trials that could test a variety of well-established therapies in COVID-19, as has REMAP-CAP1,2,3,4,5. Despite the enormous contributions of these studies, there remained an unmet need for a phase 2 mechanism for rapidly screening and triaging potential treatments for severe COVID-19 in a systematic and expedient fashion. The selection of endpoints in a clinical trial is extremely important and requires a marriage of clinical relevance with statistical reasoning.
Blinding
Although strict entry criteria may be scientifically desirable in some cases, studies with strict entry criteria may be difficult to accrue particularly when the disease is rare or alternative interventions or trials are available. Entry criteria may need to be relaxed so that enrollment can be completed within a reasonable time frame. The design of every clinical trial starts with a primary clinical research question. Secondary research questions may also be of interest but the trial design usually is constructed to address the primary research question. Information bias is when a systematic error is committed while obtaining data from the study subjects.
This is often easier than trying to make the two interventions look like each other. Participants are then randomized to receive one active treatment and one placebo (but are blinded). The downside of this approach is that the treatment schedules become more complicated (i.e., each participant must adhere to two regimens).
Ecologic study
Another concern with composite endpoints is that the interpretation can be challenging particularly when the relative importance of the components differs and the intervention effects on the components also differ. For example, how do we interpret a study in which the overall event rate in one arm is lower but the types of events occurring in that arm are more serious? Higher event rates and larger effects for less important components could lead to a misinterpretation of intervention impact. It is also possible that intervention effects for different components can go in different directions. Power can be reduced if there is little effect on some of the components (i.e., the intervention effect is diluted with the inclusion of these components).
It is much harder to design a clinical trial effectively if one doesn’t understand the interactions of genetic, environmental and/or pathophysiologic factors leading to a disease, especially one that is heterogeneous or complex (44). Success in a well-executed trial relates to good trial design, which relates to a good research question and a strong specific hypothesis, which in turn depends on a deep understanding of the subject matter and clinical research methods. For ethical reasons, any research question requires clinical equipoise (i.e. it is unclear if the intervention is better than the control) (42). No trial is thus guaranteed success, but a strong design can mitigate potential risks and augment its benefits and feasibility. One such trial applied transcutaneous electrical nerve stimulation as analgesic for PLP in 5 adult amputees (23).
Clinical study design
The design should be led by this foundation, rather than the investigator retroactively trying to find data to support a faulty foundation. The greater the supporting data, the more complex and expensive the trial design can be, and vice-versa. It is critical to gather as much preliminary data as possible and to know how to interpret it in order to choose an appropriate trial design.
Generating Clinical Trial Design Insights using Natural Language Processing - IQVIA
Generating Clinical Trial Design Insights using Natural Language Processing.
Posted: Fri, 29 Sep 2023 07:00:00 GMT [source]
Though there are various designs available, one must consider various ethical aspects of the study. Hence, each study will require thorough review of the protocol by the institutional review board before approval and implementation. However, these differences will need to be accounted during analysis of results.
Key features of clinical trials that are used to meet this objective are randomization (possibly with stratification), adherence to intent-to-treat (ITT) principles, blinding, prospective evaluation, and use of a control group. Compared to other types of study designs (e.g., case-control studies, cohort studies, case reports), randomized trials have high validity but are more difficult and expensive to conduct. Before any treatment is approved and offered to patients in the general population, rigorous evidence of its safety and efficacy must be shown. In this article, we present some general principles of good clinical trial design, which are often used as the basis to evaluate the quality of the evidence presented in manuscripts reporting trial results. By trial “design,” we include aspects from background knowledge and trial rationale to sample size and interim monitoring rules. Given that mistakes in design can seldom be later rectified, we strongly encourage investigators to consider these guidelines before beginning a study.
The CHARISMA (Bhatt et al 2006), MATCH (Diener et al 2004), and CAPRIE (Committee 1996) studies of clopidogrel for the prevention of vascular ischemic events use combinations of MI, stroke, death, and re-hospitalization as components of composite endpoints. The advantages of composite endpoints are that they may result in a more completed characterization of intervention effects as there may be interest in a variety of outcomes. Composite endpoints may also result in higher power and resulting smaller sample sizes in event-driven trials since more events will be observed (assuming that the effect size is unchanged).
Active controls and placebo controls can be used simultaneously and provide useful data. For example, if the new intervention was unable to show superiority to placebo, but an active control group was able to demonstrate superiority to placebo, then this may be evidence that the new intervention is not effective. However, if the active control with established efficacy did not demonstrate superiority to placebo, then it is possible the trial was flawed or may have been underpowered because of the placebo response or variability being unexpected high. The selection of a control group is a critical decision in clinical trial design. The control group provides data about what would have happened to participants if they were not treated or had received a different intervention. Without a control group, researchers would be unable to discriminate the effects caused by the investigational intervention from effects due to the natural history of the disease, patient or clinician expectations, or the effects of other interventions.
This can be performed in multiple ways, and one of which being as simple as a ‘flip of a coin’ to using random tables or numbers.17 The advantage of using this methodology is that it eliminates selection bias. However, the disadvantage with this methodology is that an imbalance in the number allocated to each group as well as the prognostic factors between groups. Hence, the subjects are monitored over a period of time for occurrence of a particular disease process.
This paradigm is useful only for studies with binary outcomes and are most useful when the anticipated effect size being evaluated is large. The play-the-winner and the drop-the-loser designs aim to favor the group with the best chance of success by increasing the probability of patients being randomized to that group. The probability of being randomized to one group or another is modified according to the results obtained with previous patients. The response of each patient after treatment plays an essential role in the determination of subsequent compositions of the study population.
However, occasionally the choice of trial phase (e.g. Phase II vs. Phase III) may be driven by feasibility to launch a large trial. Ultimately the design must be feasible and appropriate to answer the research question(s) of interest. A trial is only ethical if the proportion of benefit clearly outweighs the risk, especially when there are more than minimal risks. However, trials with higher potential benefits are often highly risky to patients, but may be the only existing options to help populations with devastating diseases. For instance, cancer patients undergoing investigational chemotherapy may suffer from toxic side effects, but their therapeutic options are so limited that running the study is justified.

No comments:
Post a Comment